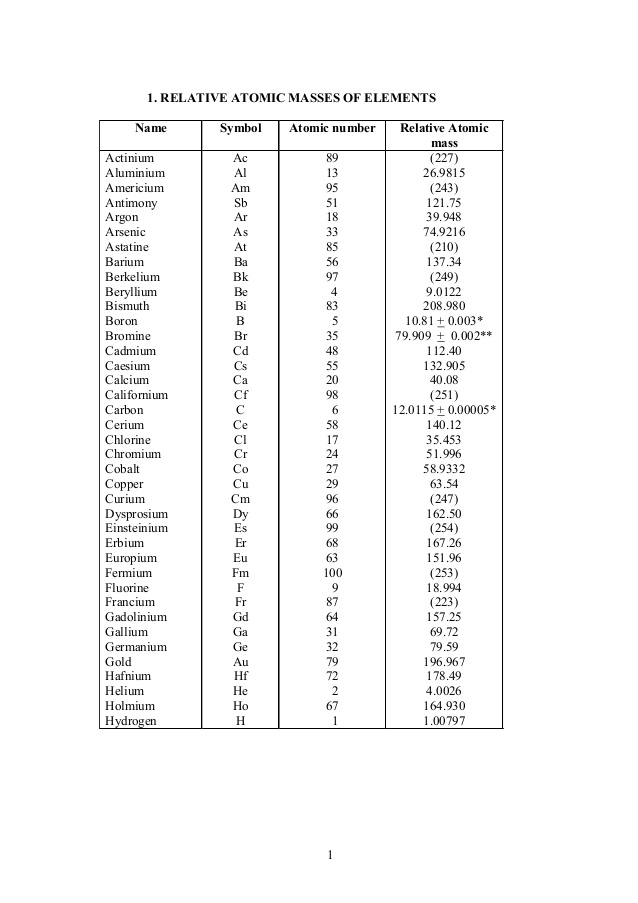
Our bismuth page has over 220 facts that span 90 different quantities. Each entry has a full citation identifying its source. Areas covered include atomic structure, physical properties, atomic interaction, thermodynamics, identification, atomic size, crystal structure, history, abundances, and nomenclature. Bismuth is a chemical element with atomic number 83 which means there are 83 protons and 83 electrons in the atomic structure. The chemical symbol for Bismuth is Bi. Bismuth is a brittle metal with a silvery white color when freshly produced, but surface oxidation can give it a pink tinge. Atomic Number: 83: Atomic Symbol: Bi: Atomic Weight: 208.9804: Electron Configuration: Xe6s 2 4f 14 5d 10 6p 3. Bismuth is the most diamagnetic of all metals.
Bismuth
| Atomic Number: | 83 | Atomic Radius: | 207 pm (Van der Waals) |
| Atomic Symbol: | Bi | Melting Point: | 271.5 °C |
| Atomic Weight: | 209.0 | Boiling Point: | 1564 °C |
| Electron Configuration: | [Xe]6s24f145d106p3 | Oxidation States: | 5, 4, 3, 2, 1, -1, -2, -3 |
Bismuth Atomic Numbers
History
From the German Weisse Masse, meaning white mass; later Wisuth and Bisemutum. In early times bismuth was confused with tin and lead. Claude Geoffroy the Younger showed it to be distinct from lead in 1753.
Properties
It is a white, crystalline, brittle metal with a pinkish tinge. It occurs in a native state. Bismuth is the most diamagnetic of all metals, and the thermal conductivity is lower than any metal, except mercury. It has a high electrical resistance, and has the highest Hall effect of any metal (i.e., greatest increase in electrical resistance when placed in a magnetic field).
Bismuth Atomic Number And Symbol
Sources
The most important ores are bismuthinite or bismuth glance and bismite. Example of avogadros law in real life. Peru, Japan, Mexico, Bolivia, and Canada are major bismuth producers. Much of the bismuth produced in the U.S. is obtained as a by-product in refining lead, copper, tin, silver, and gold ores.
Uses
Isotope Bismuth Atomic Number
'Bismanol' is a permanent magnet of high coercive force, made of MnBi, by the U.S. Naval Surface Weapons Center. Bismuth expands 3.32% on solidification. This property makes bismuth alloys particularly suited to the making of sharp castings of objects subject to damage by high temperatures. With other metals such as tin, cadmium, etc., bismuth forms low-melting alloys which are extensively used for safety devices in fire detection and extinguishing systems. Bismuth is used in producing malleable irons and is finding use as a catalyst for making acrylic fibers. When bismuth is heated in air it burns with a blue flame, forming yellow fumes of the oxide. The metal is also used as a thermocoupling material, and has found application as a carrier for 235U or 233U fuel in nuclear reactors. Its soluble salts are characterized by forming unsoluble basic salts on the addition of water, a property sometimes used in detection work. Bismuth oxychloride is used extensively in cosmetics. Bismuth subnitrate and subcarbonate are used in medicine.
